World Cancer Day 2019: Education and Inspiration Around the Globe
On February 4, 2019, organizations around the globe will come together to recognize World Cancer Day, bringing awareness to the immense burden of cancer that continues to be felt throughout the world. Joining the global community in showing support to all those affected by cancer, the American Association for Cancer Research (AACR) raises awareness about cancer and cancer research by educating the public about cancer, both in the United States and internationally.
As part of that education, the AACR recently teamed up with the Philadelphia Marathon and NBC10/Telemundo 62 in Philadelphia to present a Progress and Promise Against Cancer broadcast, bringing together some of the greatest minds in cancer research to field over 1,000 questions about cancer.
In the spirit of World Cancer Day, we’ve compiled a list of the most common questions from the public in the hopes of educating and inspiring others to raise awareness.
What can I do to lower my risk of getting cancer?
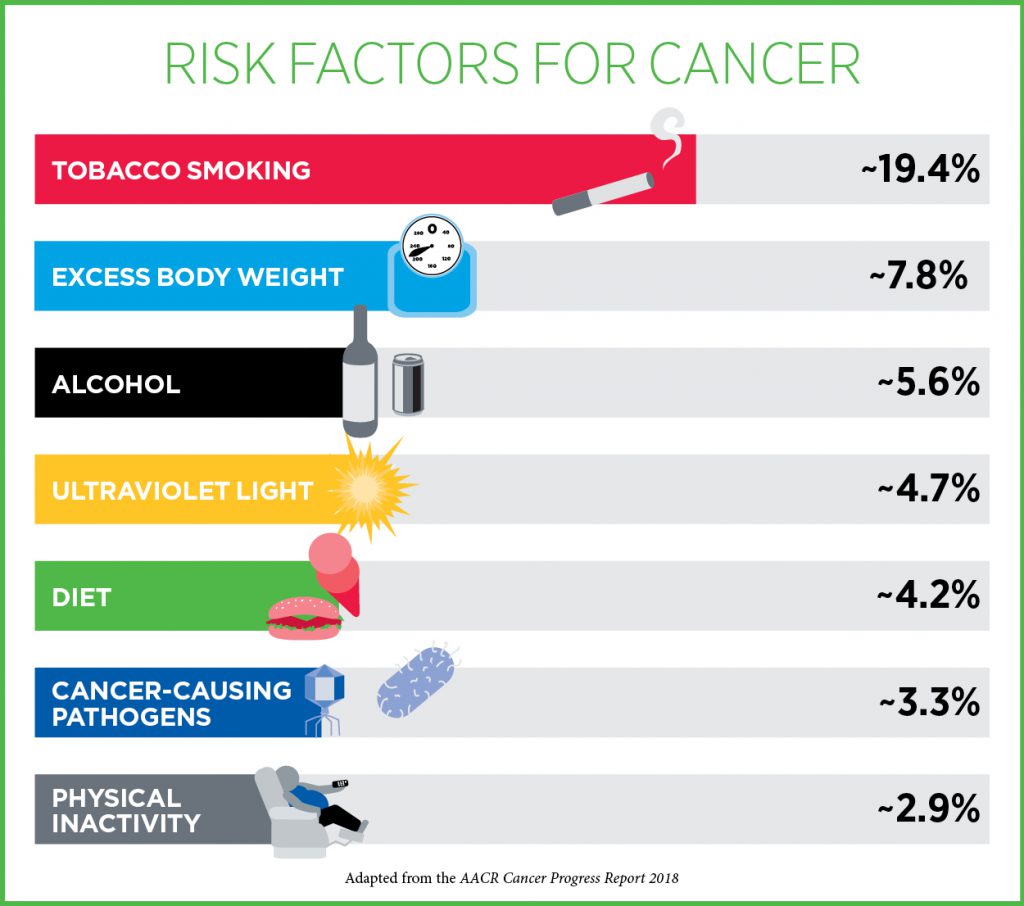
Unfortunately, there is no way to totally eliminate your risk of cancer, but researchers estimate that more than 40 percent of the cancer cases diagnosed in the United States are caused by potentially avoidable cancer risk factors. These include smoking, obesity, alcohol consumption, UV exposure, and infection with cancer-causing pathogens like human papillomavirus (HPV).
Information on preventing cancer can be found in the AACR Cancer Progress Report 2018. The Centers for Disease Prevention and Control also provides advice on cancer prevention on its website.
When and how should I be screened for cancer?
For those who are at average risk for cancer, the U.S. Preventive Services Task Force provides the following recommendations for screening:
- Breast cancer: screening mammography every two years for women ages 50 to 74, with or without clinical breast examination
- Cervical cancer: every three years with cervical cytology (a Pap smear) alone in women ages 21 to 29. For women ages 30 to 65, the USPSTF recommends screening every three years with cervical cytology alone, every five years with high-risk human papillomavirus (hrHPV) testing alone, or every five years with hrHPV testing in combination with cytology (cotesting)
- Colorectal cancer: starting at age 50 and continuing until age 75.
Some individuals are at increased risk for developing certain cancers because of factors such as age, health, and family history, and may require additional screening. In addition, it is important to keep in mind that a person’s situation may change over the course of his or her lifetime, making it vitally important that individuals continually work with their health care team to evaluate cancer screening plans.
Can cancer be detected in the blood?
Researchers have found that it is possible to detect cancer cells, cancer-cell DNA, and other cancer-cell products in the blood using tests called liquid biopsies, but most of these tests are still under development. Liquid biopsies have the potential to be safer, quicker, and more convenient for patients than a traditional tissue biopsy, which can be invasive and may not always be feasible due to the location of the tumor or other reasons. Researchers are investigating whether liquid biopsies can be used at various stages of cancer care, including for detecting cancer early, for determining a patient’s best treatment option after diagnosis, and for monitoring whether a patient’s cancer is responding to treatment or is becoming treatment resistant.
Currently, the FDA has approved only one liquid biopsy for cancer detection, a test that can be used to screen a person’s blood for a particular genetic marker associated with colorectal cancer. The other FDA-approved liquid biopsies are used for determining a patient’s best treatment option after diagnosis. You can read more about liquid biopsies in past Cancer Research Catalyst posts.
Are there any advances being made against rare or aggressive cancers?
New treatment advances are continually being made in the fight against cancer, including rare and aggressive types of cancer, as a result of research conducted in labs and clinics around the world. In 2018, the FDA approved 18 new therapeutics and expanded the use of 10 previously approved therapeutics to new types of cancer. These approvals included a molecularly targeted therapeutic for a rare type of lymphoma, as well as two targeted radiotherapeutics to treat certain rare neuroendocrine tumors. These advances are helping patients like Nicole Dicamillo, a young mom who has been living with cancer for 14 years. Thanks to the new treatment for her neuroendocrine cancer, she has seen a drastic improvement in both her scans and her quality of life. Read more about Nicole and other treatment advances in the AACR Cancer Progress Report 2018.
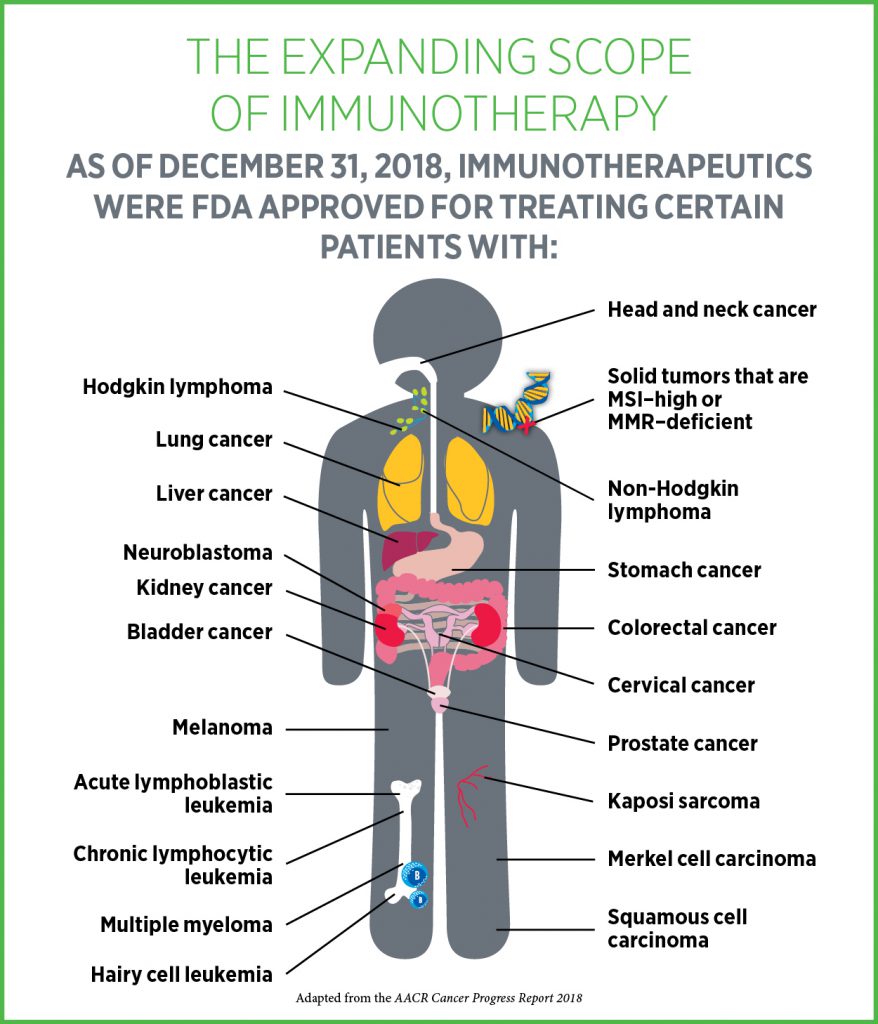
What types of cancer is immunotherapy approved for?
In the past decade, the use of immunotherapeutics in cancer has revolutionized the treatment of several cancer types. As of January 31, 2019, there were 20 immunotherapeutics approved by the FDA to treat 20 types of cancer and to treat solid tumors with a certain molecular signature.
Nobel Laureate James P. Allison, PhD, recently spoke about the future of immunotherapy in this post on Cancer Research Catalyst, predicting that the use of immunotherapy will continue to expand over the next year and beyond, mostly by combining immunological agents, or using them in combination with chemotherapy, radiation, and molecularly targeted drugs.
To join the movement on World Cancer Day, visit WorldCancerDay.org. Participants are encouraged to take action against cancer by spreading the message on social media, sharing their own cancer story, and empowering the next generation to raise awareness. These are all steps in the right direction toward the goal of preventing and curing all cancers.