New Therapeutic Approved by FDA to Treat Breast Cancer
Earlier this week, the U.S. Food and Drug Administration (FDA) approved the molecularly targeted therapeutic ribociclib (Kisqali) for use in combination with any aromatase inhibitor for initial treatment of postmenopausal women with a specific subtype of breast cancer: hormone receptor (HR)–positive, HER2-negative, advanced breast cancer.
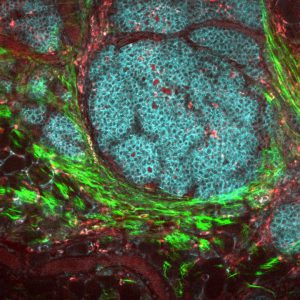
The image of a breast cancer tumor and its microenvironment was obtained using multiphoton microscopy and endogenous fluorescence. Image courtesy of the National Cancer Institute.
Cancer arises when the orderly processes that control the multiplication and life span of normal cells go awry. For the approximately three in every four breast cancers diagnosed in the United State that are HR-positive, cancer cell multiplication is fueled by the hormones estrogen and/or progesterone.
Most postmenopausal women diagnosed with advanced HR-positive, HER2-negative breast cancer are treated with antihormone therapies such as an aromatase inhibitor. However, most of these women eventually have disease recurrence as their cancers become resistant to these therapeutics.
Ribociclib is a molecularly targeted therapeutic that blocks the function of two specific proteins that play a role in driving cell multiplication—cyclin-dependent kinase 4 (CDK4) and CDK6. Therefore, researchers set out to test whether adding ribociclib to aromatase inhibitor therapy could delay or prevent the emergence of resistance.
Results from the phase III MONALEESA-2 clinical trial that led to this week’s approval of ribociclib showed that the combination did indeed delay disease progression for postmenopausal women with HR-positive, HER2-negative advanced breast cancer. Specifically, the results, which were published late last year in The New England Journal of Medicine, showed that after 18 months, the progression-free survival rate was 63.0 percent among those randomly assigned ribociclib and letrozole and 42.2 percent among those randomly assigned placebo and letrozole. In addition, the estimated median progression-free survival was 14.7 months for those in the placebo-containing arm and had not been reached for the ribociclib-containing arm.
Ribociclib is the second CDK4/6 inhibitor to be approved by the FDA for use as part of initial treatment for postmenopausal women with advanced HR-positive, HER2-negative breast cancer. As discussed on this blog previously, the first CDK4/6 inhibitor—palbociclib (Ibrance)—was approved for this indication in February 2015; the ability of this molecularly targeted therapeutic to transform the lives of patients for the better is highlighted by the experience of Janet Klein.
To learn more about how Janet is living a good life with stage 4 breast cancer thanks to palbociclib, read her story in the AACR Cancer Progress Report 2015 or watch the video below.
https://www.youtube.com/watch?v=00JJVQCeGAk