AACR Annual Meeting 2016: Research, Prevention, and the Affordable Care Act
During a conference known for its emphasis on basic and translational science, a panel discussion on the Affordable Care Act (ACA) and its effects on research and prevention filled a packed room at the American Association for Cancer Research (AACR) Annual Meeting 2016 in New Orleans.
“I don’t usually see a policy discussion at the AACR meeting, nor one that is so well attended,” said cancer researcher and panelist Ernest T. Hawk, MD, MPH, co-chairperson of the AACR’s Cancer Prevention Committee and vice president and division head for Cancer Prevention and Population Sciences at the University of Texas MD Anderson Cancer Center in Houston, as he looked over at the crowded room where dozens of people lined up along the walls and sat on the floor.
Mary Jackson Scroggins, a patient advocate and ovarian cancer survivor, opened the session with her own type of “nonscientific research.” Scroggins conducted a web-based survey and also talked to people on the bus in her home city of Washington, D.C., to learn what they knew about the Affordable Care Act (ACA), often referred to as “Obamacare.”
Responses varied: Some were happy to finally have health insurance while others struggled to pay high premiums. Other responses were more comical: Scroggins told of one elderly woman who had just gotten health insurance through the ACA marketplace and stated, “I’m so glad I don’t have Obamacare.”
“There are lots of misconceptions—even about the name,” said Scroggins, who noted that responses changed when she used the full name of the legislation: the Patient Protection and Affordable Care Act. “When you add the patient protection part, people have no idea what it means.”
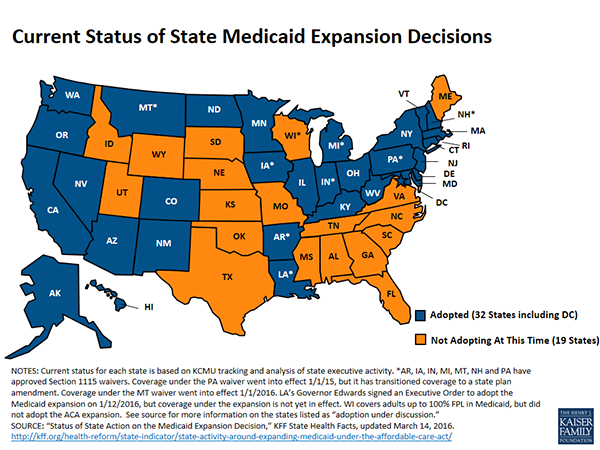
Not everyone has access to health care coverage afforded under the law, specifically in the 19 states that have refused to expand their Medicaid programs, said panelist Jennifer Singleterry, a senior analyst with the American Cancer Society Cancer Action Network (ACS CAN) in Washington, D.C. In fact, 56 percent of the 3,400 people who contacted ACS’ health insurance assistance call center in 2015 lived in states that did not expand their Medicaid programs.
“These people were too rich for Medicaid, too poor for the marketplace, and had a lack of health literacy to seek existing programs,” said Singleterry.
She also noted a growing cost concern specific to cancer patients and those facing serious disease: Many insurance plans require patients to pay co-insurance, which is a percentage of a drug’s cost, instead of a co-pay, a flat amount no matter how much the drug costs. Thus, patients often are unsure what their final drug costs will be—and with cancer drugs sometimes costing thousands of dollars, this price tag can be significant. In addition, many cancer drugs, even generics, fall in the top tier of the formulary, which means patients must pay more, Singleterry said.
An ACS CAN 2014 analysis of health insurance plans available through the marketplace also showed 43 percent of health plans did not provide out-of-network coverage—a literal lifeline for someone who may want doctors with specific experience, particularly in the cases of rare or difficult-to-treat cancers. In eight states, fewer than 20 percent of health care marketplace plans offered out-of-network coverage, Singleterry said. She told the story of a cancer survivor who was suspected of having a recurrence of her cancer, but her health insurance plan didn’t include even one in-network oncologist. The ACS is helping the woman get care, but this is obviously causing “a lot of extra stress that she doesn’t need right now,” Singleterry said.
The ACA also has provisions mandating insurance coverage, in most cases, for the cost of routine care for patients participating in clinical trials. However, a study showed that close to 64 percent of 264 cancer centers and research programs reported receiving denials of insurance coverage for routine care for patients in clinical trials, said panelist William S. Dalton, MD, PhD, who was not involved in the study. Dalton heads M2Gen, a Moffitt Cancer Center biotechnology company, and is the founding director of Moffitt’s Personalized Medicine Institute based in Tampa, Florida.
Another cornerstone of the ACA is prevention. “Prevention is clearly an important premise of this act,” said Hawk. In 2015, 137.7 million people had access to free preventive screening—up from 71 million in 2011 and 2012, he noted. In addition, some studies show an uptick in the number of people taking preventive cancer measures, such as in rates of women receiving HPV vaccinations and men getting colonoscopies.
One study showed that colonoscopy screening rates of men ages 66-75 increased 4 percent in 2015 compared with previous years, though the study did not show the same uptick for women. Most notable is that screening rates increased 9.2 percent among men reporting cost as a barrier to medical care and 7.5 percent among the least educated, Hawk said.
Noting that just 3 percent of health care spending is dedicated to prevention and public health and that 75 percent of health care costs are related to preventable conditions, Hawk explained that the ACA also established a mandatory prevention fund that set aside $15 billion over 10 years starting in 2010 to help address these issues. However, the $2 billion originally allocated for 2015 was cut to $927 million, Hawk noted, and more cuts are scheduled.
Panelists also discussed other areas of research covered by the legislation. For example, the ACA established the Patient-Centered Outcomes Research Institute (PCORI), a nonprofit institution tasked with measuring the effectiveness of treatments and strategies, Dalton explained. In addition, the legislation established the Cures Acceleration Network, charged with translating basic science so it can benefit patients. Both organizations provide grants for research studies.
This post is adapted from an article on the website of Cancer Today, a quarterly magazine for cancer patients, survivors, and caregivers published by the AACR. Read all of Cancer Today’s AACR Annual Meeting 2016 coverage.